| Availability: | |
|---|---|
| Quantity: | |
DINGMIAO
Product Show
The use of botulinum toxin type A to reduce the volume of muscles such as masseter, temporalis, calf, and deltoid is becoming more popular and promise to have increasingly broader applications.


Product Usage
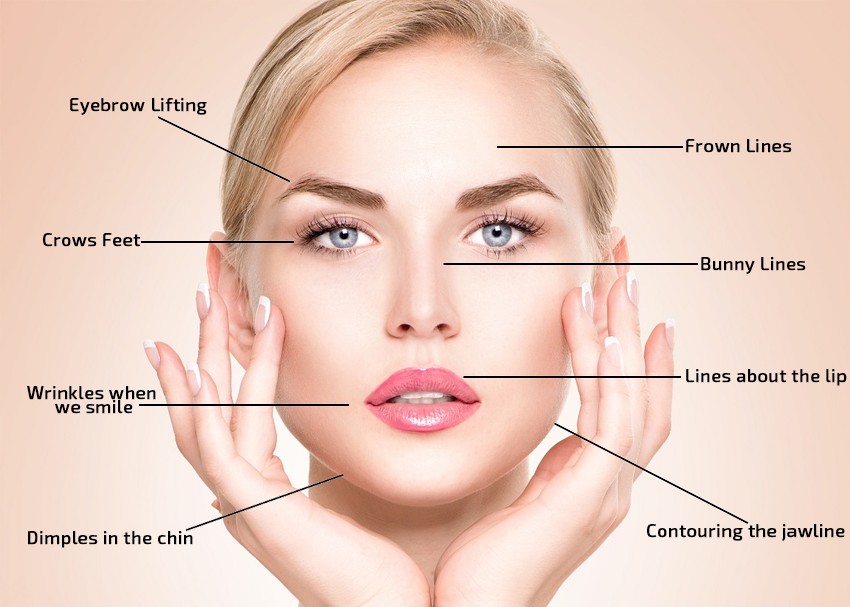
Meditoxin 100u (Type A) Similarly to Botulinum Toxin , temporarily corrects certain types of medium to deep facial wrinkles by relaxing the muscles into which it was applied. This property also makes it suitable not only for therapeutic cosmetic procedures but also for medical treatment of various conditions among which are blepharospasm, TMJ, stroke recovery and many more.
Product Advantages
Resume normal work right away
Just by few tiny injections
Effects visible after one administration, starting from 2 weeks after injection, and peaks around 3~4 weeks.
Painless even when used without aenesthetic
Effects last for 6~12 months
Approved by Health Authorities with over 8 years’ clinical practices.
Rug Effects
After Injection
There is the possibility of reactions at the site of an injection, and effects beyond the injection site, including but not limited to asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. Serious hypersensitivity has been reported as rare.
There is a caution for people with pre-existing neuromuscular disorders, and for those with corneal exposure and ulceration in blepharospasm.
Botulium Toxin Type A is not to be administered at all if a patient has a known allergy to any of the ingredients, or if the patients have neuromuscular junctional disorders or known to have adverse reactions.
To preserve results achieved after a treatment with this Botulinum Toxin Type A injectable, patients need to undergo maintenance injection procedure
Other Products
 Hipretty PDO Thread |  Younsofill Dermal Fillers |
 Cindella + Luthione + VitaminC |  Lipolab |  LIporase |
Product Show
The use of botulinum toxin type A to reduce the volume of muscles such as masseter, temporalis, calf, and deltoid is becoming more popular and promise to have increasingly broader applications.


Product Usage

Meditoxin 100u (Type A) Similarly to Botulinum Toxin , temporarily corrects certain types of medium to deep facial wrinkles by relaxing the muscles into which it was applied. This property also makes it suitable not only for therapeutic cosmetic procedures but also for medical treatment of various conditions among which are blepharospasm, TMJ, stroke recovery and many more.
Product Advantages
Resume normal work right away
Just by few tiny injections
Effects visible after one administration, starting from 2 weeks after injection, and peaks around 3~4 weeks.
Painless even when used without aenesthetic
Effects last for 6~12 months
Approved by Health Authorities with over 8 years’ clinical practices.
Rug Effects
After Injection
There is the possibility of reactions at the site of an injection, and effects beyond the injection site, including but not limited to asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence and breathing difficulties. Serious hypersensitivity has been reported as rare.
There is a caution for people with pre-existing neuromuscular disorders, and for those with corneal exposure and ulceration in blepharospasm.
Botulium Toxin Type A is not to be administered at all if a patient has a known allergy to any of the ingredients, or if the patients have neuromuscular junctional disorders or known to have adverse reactions.
To preserve results achieved after a treatment with this Botulinum Toxin Type A injectable, patients need to undergo maintenance injection procedure
Other Products
 Hipretty PDO Thread |  Younsofill Dermal Fillers |
 Cindella + Luthione + VitaminC |  Lipolab |  LIporase |